Neckel Lab
Analysing the Signalling Cascades in the Enteric Nervous System (ENS)
Our work primarily focuses on understanding how the enteric nervous system (ENS) works. Aside the brain, the ENS is the largest and arguably most complex neural network found within the body. With approximately 500 million neurons and four to ten times as many enteric glial cells, it forms ganglionated plexus within the walls of the gastrointestinal tract. These plexus exhibit a remarkable histoarchitectural complexity and can regulate many vital intestinal functions even when disconnecting the organ from the rest of the body. Thus, the ENS is often referred to as brain in the gut or second brain.
Our current interests are cantered at unravelling molecular signals and intercellular communication that drive the cellular homeostasis and the development of the ENS in postnatal life. Therefore, we work with murine reporter models as well as human patient- and post-mortem specimens to investigate the influence of morphogens, such as Wnt, R-Spondin, Dkk or matrix molecules on the proliferative capacity and neuronal differentiation of ENS progenitor cells. Moreover, we analyse the expression pattern of differentiation- and functional markers in patient samples to improve our understanding of the pathomechanistic basis of enteric neuropathies like Hirschsprung’s disease or gastroschisis-related dysmotility.
Contact
Dr rer. nat. Peter Neckel
Working Group Leader | Head of Body Donation Programme
Key Methods of the Lab
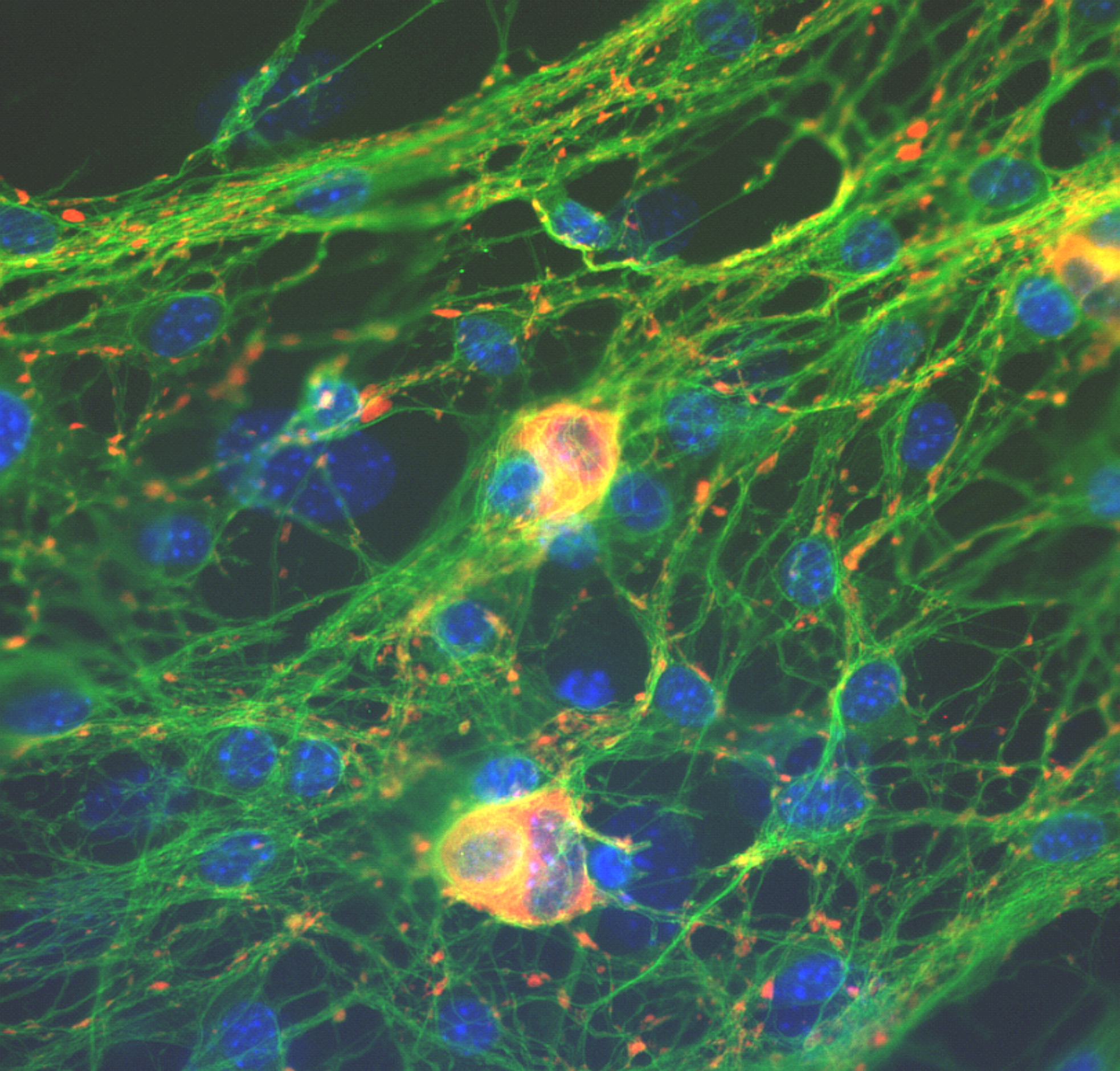
Primary cell culture of murine and human ENS cells, organotypic culture systems of intestinal tissues, proliferation and neurogenesis assays, immunohistochemistry, tissue clearing.
Future directions of the Lab
Wnt/R-Spondin/LGR – Molecular pathways regulating ENS progenitor cells
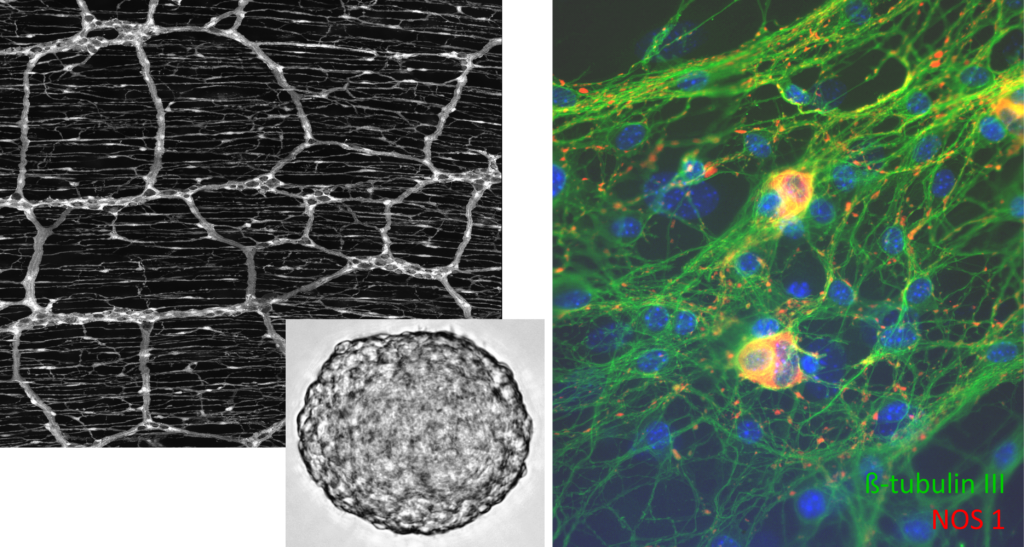
Ongoing and future projects focus on the involvement of the Wnt-regulatory network in the proliferation and neural differentiation of enteric neural progenitor cells, particularly in the postnatal ENS. We found that activation of the canonical Wnt pathway increased the yield of newly generated neurons in in vitro cultures of murine and human, patient-derived ENS progenitor cells. Genome-wide gene expression analyses also revealed that the canonical Wnt pathway is involved in switching ENS progenitor cells from proliferative programs to differentiation. In current experiments, we are looking into the role of R-Spondins, Wnt-activators known for their regulatory profile in other stem cell compartments, in fate decision of ENS progenitor cells.
Neurogenesis and the Aging ENS
Although the regenerative potential of the ENS is very limited in vivo, a subpopulation of ENS cells maintain the potential to proliferate and form new neurons. These ENS progenitors can be isolated from murine and human intestines even at higher ages and exhibit an astonishing neurogenic potential in vitro. In ongoing experiments, we want to map the proliferative and neurogenic potential of ENS progenitors and assess age-related changes. Additionally, we collect and analyse human intestinal specimens from paediatric, adolescent, middle aged, and elderly patients as well as post-mortem samples from body donors to evaluate changes of the aging ENS in humans.
Mechanotransduction and homeostasis in the ENS
The enteric nervous system is constantly in movement. The intestinal peristalsis permanently compresses and stretches the enteric ganglia exerting tremendous mechanical forces onto neurons and glial cells. In this future project, we aim to elucidate how ENS cells sense and react to mechanical stimulation. Using various passive and active mechanical cues, we want to elucidate the molecular signalling pathways, such as the Hippo-YAP/TAZ cascade, involved in mechanotransduction. Moreover, we want to find out, how intestinal motility shapes ENS homeostasis and function.
Selected Publications
Click here for a full list of publications.
- M Scharr, S Scherer, B Hirt, PH Neckel. Dickkopf1 induces enteric neurogenesis and gliogenesis in vitro if apoptosis is evaded. Commun Biol. 2023 Aug 2;6(1):808. DOI: 10.1038/s42003-023-05072-x.
- Y Zhang, K Seid, F Obermayr, L Just, PH Neckel. Activation of Wnt Signaling Increases Numbers of Enteric Neurons Derived From Neonatal Mouse and Human Progenitor Cells. Gastroenterology. 2017 153:154-165 e159. DOI: 10.1053/j.gastro.2017.03.019
- PH Neckel, M Scharr, K Seid, K Nothelfer, J Fuchs, F Obermayr, B Hirt, SM Huber, L Just. Wnt receptor Frizzled-4 as a marker for isolation of enteric neural progenitors in human children. Cells. 2019, 8(8), 792. DOI: 10.3390/cells8080792
- PH Neckel, U Mattheus, B Hirt, L Just, AF Mack. Large-scale tissue clearing (PACT): Technical evaluation and new perspectives in immunofluorescence, histology, and ultrastructure. Sci Rep. 2016 Sep 29;6:34331. DOI: 10.1038/srep34331
- F Deffner, M Scharr, S Klingenstein, M Klingenstein, A Milazzo, S Scherer, A Wagner, B Hirt, AF Mack, PH Neckel. Histological evidence for the enteric nervous system and the choroid plexus as alternative routes of neuroinvasion by SARS-CoV2. Front. Neuroanat. 2020 Oct 6;14:596439. DOI: 10.3389/fnana.2020.596439
Lab Members
Dr rer. nat. Peter Neckel
Working Group Leader | Head of Body Donation Programme
Dr. Simon Scherer
Clinician Scientist
Jon Nicola van den Beld
Medical PhD Student | IZKF-Promotionskolleg Beneficiary
Melanie Scharr, M. Sc.
Scientific PhD Student
Nicole Schlecht
Medical PhD Student | IZKF-Promotionskolleg Beneficiary
Sophia Ritter
Medical PhD Student | IZKF-Promotionskolleg Beneficiary
The Lab on Social Media
Follow us on Twitter/X:
@NeckelLab
